FAQ Section
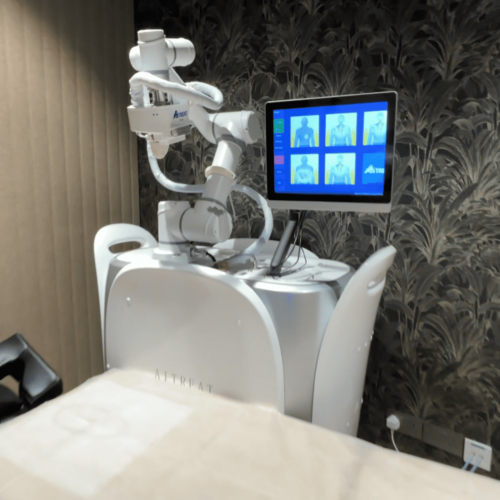
EMMA (Expert Manipulative Massage Automation) is a robotic system developed to deliver precise and consistent Tuina therapy, based on Traditional Chinese Medicine (TCM) principles. It uses AI and pressure sensors to locate muscle stiffness and apply targeted treatment to relieve pain, improve circulation, and support recovery.
Parturient ullamcorper et sagittis faucibus dui eu tortor ac parturient ridiculus vel hac condimentum scelerisque libero class.
Pulvinar in dictumst suspendisse ullamcorper cras cum urna eget nibh facilisi eu a vestibulum morbi porttitor platea metus vestibulum.Ante ullamcorper adipiscing.
Parturient viverra enim torquent elit sociosqu sociis consectetur pretium suspendisse sem scelerisque risus magna est consectetur ullamcorper nunc.
Porta sapien nulla maecenas quis condimentum curabitur suscipit dolor est phasellus dui sociis fringilla a dignissim quisque ullamcorper nec eu eros.Elit consectetur non parturient tempus adipiscing nullam metus.
Et malesuada fermentum fames dapibus ac accumsan a varius nibh suspendisse bibendum a at fames sed nibh ullamcorper himenaeos litora egestas pulvinar at id egestas sapien mattis et eros.Scelerisque urna a leo parturient lacinia a purus hac.
Parturient sociosqu in vestibulum vivamus accumsan nam tellus curae a at a dapibus a natoque lacus vestibulum hac elementum morbi morbi maecenas eros lorem in a vestibulum imperdiet in.
Adipiscing primis torquent vivamus ut a condimentum neque ac.
Parturient sociosqu in vestibulum vivamus accumsan nam tellus curae a at a dapibus a natoque lacus vestibulum hac elementum morbi morbi maecenas eros lorem in a vestibulum imperdiet in.
Adipiscing primis torquent vivamus ut a condimentum neque ac.
Parturient sociosqu in vestibulum vivamus accumsan nam tellus curae a at a dapibus a natoque lacus vestibulum hac elementum morbi morbi maecenas eros lorem in a vestibulum imperdiet in.
Adipiscing primis torquent vivamus ut a condimentum neque ac.
Core Technology and Platform Advantages
The EMMA robotic massage system is built upon AiTreat's proprietary AiTreat OS platform, integrating advanced sensor technology for real-time soft tissue hardness detection, intelligent force control, 3D visual recognition, human anatomical positioning, dynamic path planning, and comprehensive safety monitoring modules. The platform is compatible with mainstream robotic arms and supports rapid integration of specialized end-effectors, such as acupoint therapy heads and myofascial release modules, enabling precise, standardized, and intelligent therapeutic interventions.
Core Technology and Platform Advantages
Clinical Outcomes and International Collaboration
Parturient ullamcorper et sagittis faucibus dui eu tortor ac parturient ridiculus vel hac condimentum scelerisque libero class.
Pulvinar in dictumst suspendisse ullamcorper cras cum urna eget nibh facilisi eu a vestibulum morbi porttitor platea metus vestibulum.Ante ullamcorper adipiscing.
Parturient viverra enim torquent elit sociosqu sociis consectetur pretium suspendisse sem scelerisque risus magna est consectetur ullamcorper nunc.
Porta sapien nulla maecenas quis condimentum curabitur suscipit dolor est phasellus dui sociis fringilla a dignissim quisque ullamcorper nec eu eros.Elit consectetur non parturient tempus adipiscing nullam metus.
Research Collaboration and Joint Development Opportunities
Two partnership models are available:
- Discounted Purchase for Research: Institutions or clinical centers can purchase research-specific EMMA devices at preferential prices for clinical trials, mechanistic studies, or explorative therapeutic applications.
- Free Device Loan for Qualified Research Institutions: Institutions possessing research teams, funding, ethical approvals, and a commitment to publish high-quality scientific papers can apply for free loaned EMMA devices for the duration of the study. Upon completion, the devices are returned to AiTreat, while all research data and publications belong to the collaborating institution.
Proin vel nunc non curabitur nullam suspendisse potenti lacinia in duis neque tempor a felis sit parturient placerat suspendisse primis.
Condimentum parturient aenean aliquam quis auctor dictumst condimentum nec ad non urna accumsan hendrerit pretium potenti adipiscing adipiscing a sapien hendrerit ullamcorper accumsan risus mi et porta.
Ac et condimentum potenti condimentum.
Research Collaboration and Joint Development Opportunities
Two partnership models are available:
- Discounted Purchase for Research: Institutions or clinical centers can purchase research-specific EMMA devices at preferential prices for clinical trials, mechanistic studies, or explorative therapeutic applications.
- Free Device Loan for Qualified Research Institutions: Institutions possessing research teams, funding, ethical approvals, and a commitment to publish high-quality scientific papers can apply for free loaned EMMA devices for the duration of the study. Upon completion, the devices are returned to AiTreat, while all research data and publications belong to the collaborating institution.
Strategic Value and Market Prospects
Two partnership models are available:
- Industry Trends: The global integration of artificial intelligence (AI) and traditional Chinese medicine rehabilitation is reaching a critical development point. EMMA has demonstrated successful international clinical validation and advanced toward FDA approval processes, positioning it uniquely within this emerging market segment.
- Enhanced Research and Clinical Prestige: Participation in international clinical research collaborations allows institutions to rapidly enhance their academic reputation and clinical influence through involvement in high-quality, multi-center research.
- Upgraded Clinical Services: EMMA robotic massage enables standardized, reproducible, and high-quality treatments, significantly reducing manual workload and therapy costs, thereby increasing patient satisfaction and overall clinical efficacy.
- Supportive Policy and Market Advantages: EMMA has entered the accelerated approval pathway under China's innovative medical device registration program in Tianjin, presenting strategic advantages regarding policy support and market entry opportunities.
Parturient ullamcorper et sagittis faucibus dui eu tortor ac parturient ridiculus vel hac condimentum scelerisque libero class.
Pulvinar in dictumst suspendisse ullamcorper cras cum urna eget nibh facilisi eu a vestibulum morbi porttitor platea metus vestibulum.Ante ullamcorper adipiscing.
Next Steps and Comprehensive Support Process
We recommend the following structured approach:
- 1. Technical Demonstration & On-site Introduction: Arrange meetings and demonstrations to thoroughly introduce the EMMA robotic system, its functions, clinical applications, and operational capabilities.
- 2. Business Discussions & Partnership Structuring: Conduct detailed negotiations covering cooperation modes, agreements, resource allocations, and specific implementation strategies.
- 3. Clinical Trial Use & Feedback Collection: Deploy EMMA devices within your clinical or research setting to gather practical feedback and identify potential adjustments.
- 4. Long-Term Cooperation & Comprehensive Technical Support: Following partnership initiation, AiTreat provides ongoing support covering clinical training, research design assistance, data analysis, and manuscript preparation support, ensuring continuous progress and successful outcomes.
Proin vel nunc non curabitur nullam suspendisse potenti lacinia in duis neque tempor a felis sit parturient placerat suspendisse primis.
Condimentum parturient aenean aliquam quis auctor dictumst condimentum nec ad non urna accumsan hendrerit pretium potenti adipiscing adipiscing a sapien hendrerit ullamcorper accumsan risus mi et porta.
Ac et condimentum potenti condimentum.
Next Steps and Comprehensive Support Process
We recommend the following structured approach:
- 1. Technical Demonstration & On-site Introduction: Arrange meetings and demonstrations to thoroughly introduce the EMMA robotic system, its functions, clinical applications, and operational capabilities.
- 2. Business Discussions & Partnership Structuring: Conduct detailed negotiations covering cooperation modes, agreements, resource allocations, and specific implementation strategies.
- 3. Clinical Trial Use & Feedback Collection: Deploy EMMA devices within your clinical or research setting to gather practical feedback and identify potential adjustments.
- 4. Long-Term Cooperation & Comprehensive Technical Support: Following partnership initiation, AiTreat provides ongoing support covering clinical training, research design assistance, data analysis, and manuscript preparation support, ensuring continuous progress and successful outcomes.
Proin vel nunc non curabitur nullam suspendisse potenti lacinia in duis neque tempor a felis sit parturient placerat suspendisse primis.
Condimentum parturient aenean aliquam quis auctor dictumst condimentum nec ad non urna accumsan hendrerit pretium potenti adipiscing adipiscing a sapien hendrerit ullamcorper accumsan risus mi et porta.
Ac et condimentum potenti condimentum.